As you want to get information about Entrance test for MSc Chemistry admission in NIT Calicut so here I am giving you same:
NIT Calicut MSc Chemistry Entrance Exam Eligibility:
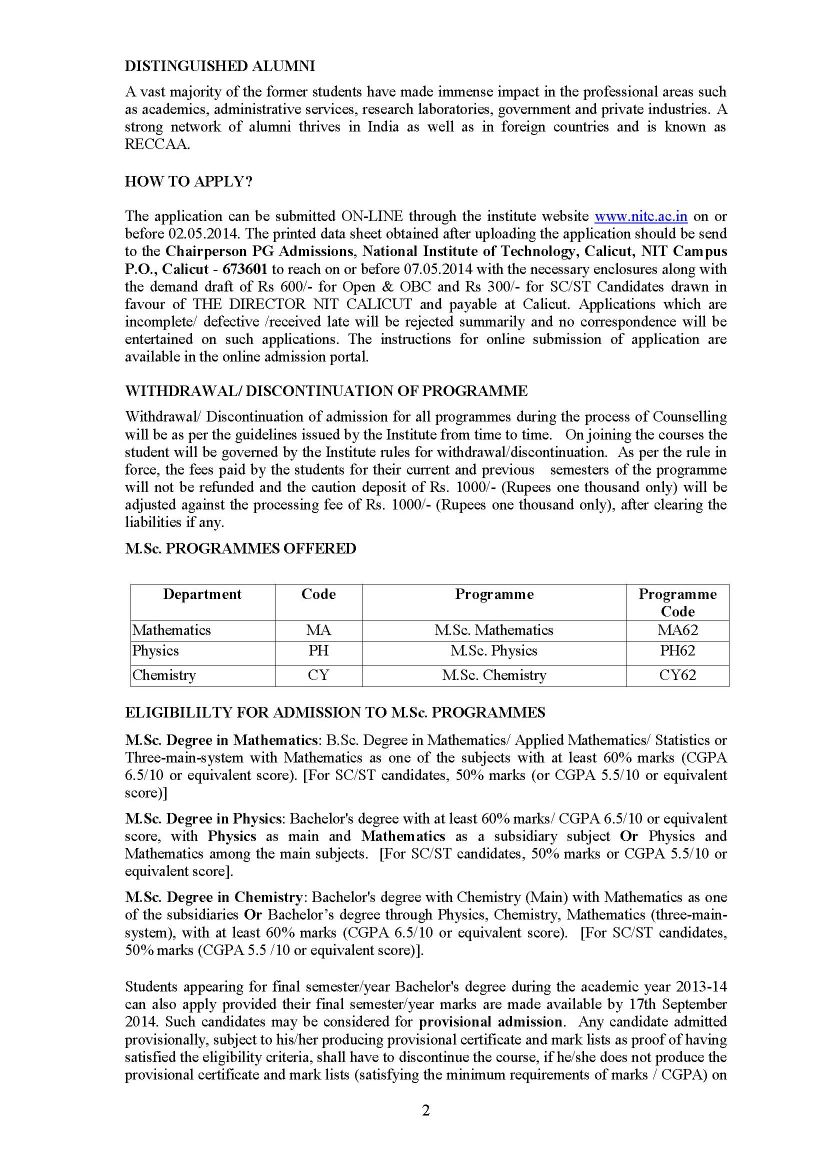
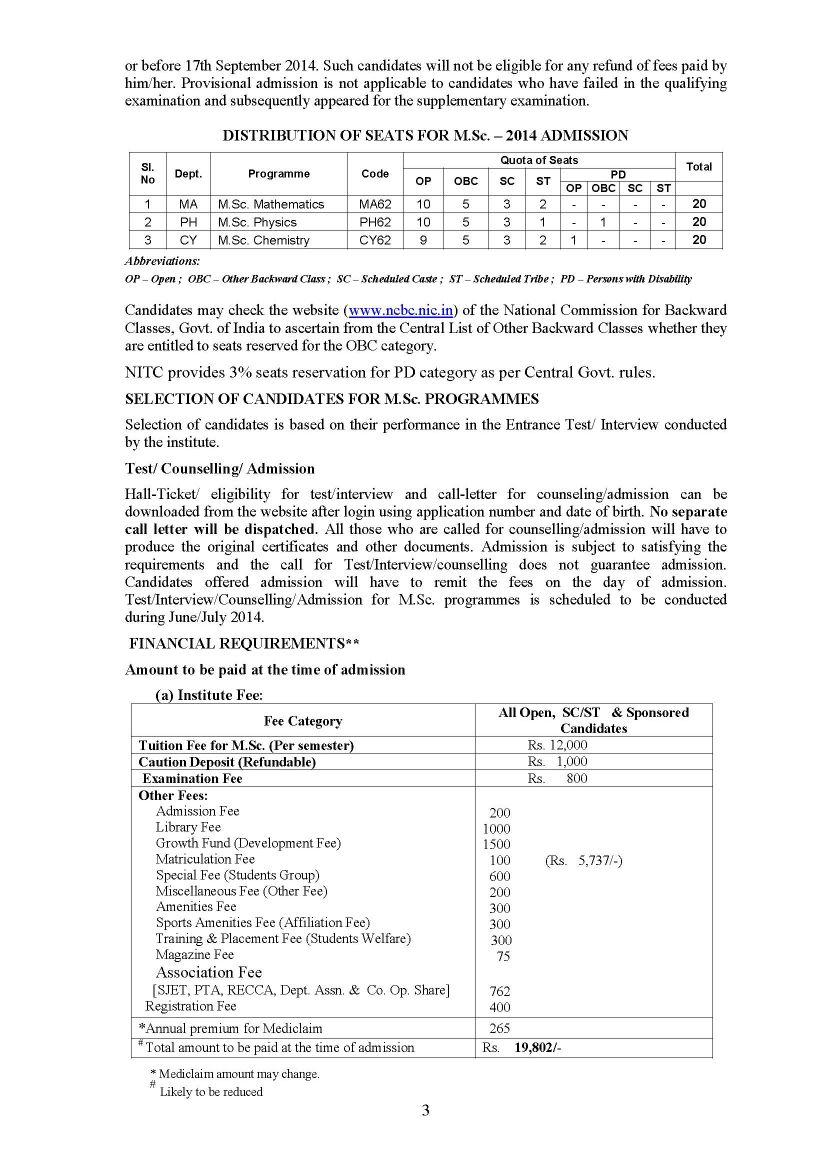
Candidate must have bachelor’s degree with Chemistry (Main) with Mathematics as one of the subsidiaries Or Bachelor’s degree through Physics, Chemistry, Mathematics (three-main system), with at least 60% marks.
Application Form Fee:
Rs 600/- for General and Open & OBC Candidates
Rs 300/- for SC/ST Candidates
Entrance Exam Syllabus:
Theoretical Chemistry: Bohr’s theory of hydrogen atom, Wave-particle duality, Uncertainty
principle, Schrödinger’s wave equation, Quantum numbers, shapes of orbitals, Hund’s rule,
Aufbau principle and Pauli exclusion principle, Types of bonding, Orbital theory of covalency,
Hydrogen bonding, Valence bond theory and molecular orbital theory, VSEPR theory and shapes
of molecules, Hybridization,
Physical Chemistry: Kinetic theory of gases, Types of molecular velocities, Mean free path,
Principle of equipartition of energy, First, second and third law of thermodynamics and its
applications, thermochemistry, Kirchhoff equation, Maxwel’s relationships, Criteria for
Spontaneity, Clausius-Clapeyron equation, Le Chatlier Principle, Ionic equillibria in solutions,
pH and buffer solutions, Hydrolysis, Solubility product, Colligative Properties, Phase rule and
Phase diagrams of one and two-component systems, Electrochemistry-Conductance and its
applications, Ionic mobility, Transport number, Kohlrausch’s law, Ionic strength, Galvanic cells,
Nernst equation, EMF and free energy, Polarography, Chemical kinetics-Rate equation, Half life,
Order and molecularity, Arrhenius equations, Enzyme kinetics, Photophysical and photochemical
processes, Spectroscopy-IR, UV-Visible, NMR and Mass spectroscopy.
Organic Chemistry: Basic concepts in organic chemistry, Isomerism and nomenclature,
Electronic effects, Optical isomerism, Designation of absolute configuration, Conformational
isomers, Reactions involving stereoisomers, Aromaticity and Huckels rule, Preparation and
reactions of alkanes, alkenes, alkynes, arenes, and their functional derivatives, Electrophilic
aromatic substitution, Nucleophilic substitution and elimination reactions, Addition reactions,
Rearrangements, Grignard reagents, Acetoacetic ester and Malonic ester chemistry, Alkaloids,
Terpenes, Carbohydrates, Amino acids, Peptides, Nucleic acids, Heterocyclic chemistry.
Inorganic Chemistry: Periodicity in atomic properties, Methods of isolation and purification of
elements, Main group Elements (s and p block)-General characteristics, Structure of electron
deficient compounds,Transition and inner transition metals (d and f Block)-Coordination
complexes-Nomenclature, Stereochemistry and isomerism, Stability of complexes,
VB theory,
crystal field theory and MO theory: Hybridization, Crystal field splitting, Magnetic properties and
colour of metal complexes. Lanthanide and actinide series, Structure of solids-NaCl and CsCl,
Lattice energy in ionic solids, Crystal defects, Nuclear chemistry-Fundamental particles of the
nucleus, Nuclear binding energy, Artificial radioactivity, Analytical chemistry-Principles of
qualitative, quantitative and gravimetric analysis, Thermal analytical methods, Separation and
purification techniques.
NIT Calicut MSc Chemistry Entrance Exam Syllabus and Questions