|
#4
23rd February 2015, 08:48 AM
| |||
| |||
| Re: CBSE Latest Sample Papers for Class 12th Chemistry
Below I am providing you some questions of from question paper of CBSE (Central Board of Secondary Education) for Class 12th Chemistry and attaching a PDF attachment which has detail information regarding this that you can down load for free: Give the formula of a noble gas species which is isostructural with IBr2 What is the effect of synergic bonding interactions in a metal carbonyl complex? PCl5 acts as an oxidizing agent. Justify. Write the name of the product formed when benzenediazonium chloride solution is treated with potassium iodide. Name the crystal defect which reduces the density of an ionic solid? What type of ionic substances show this defect? 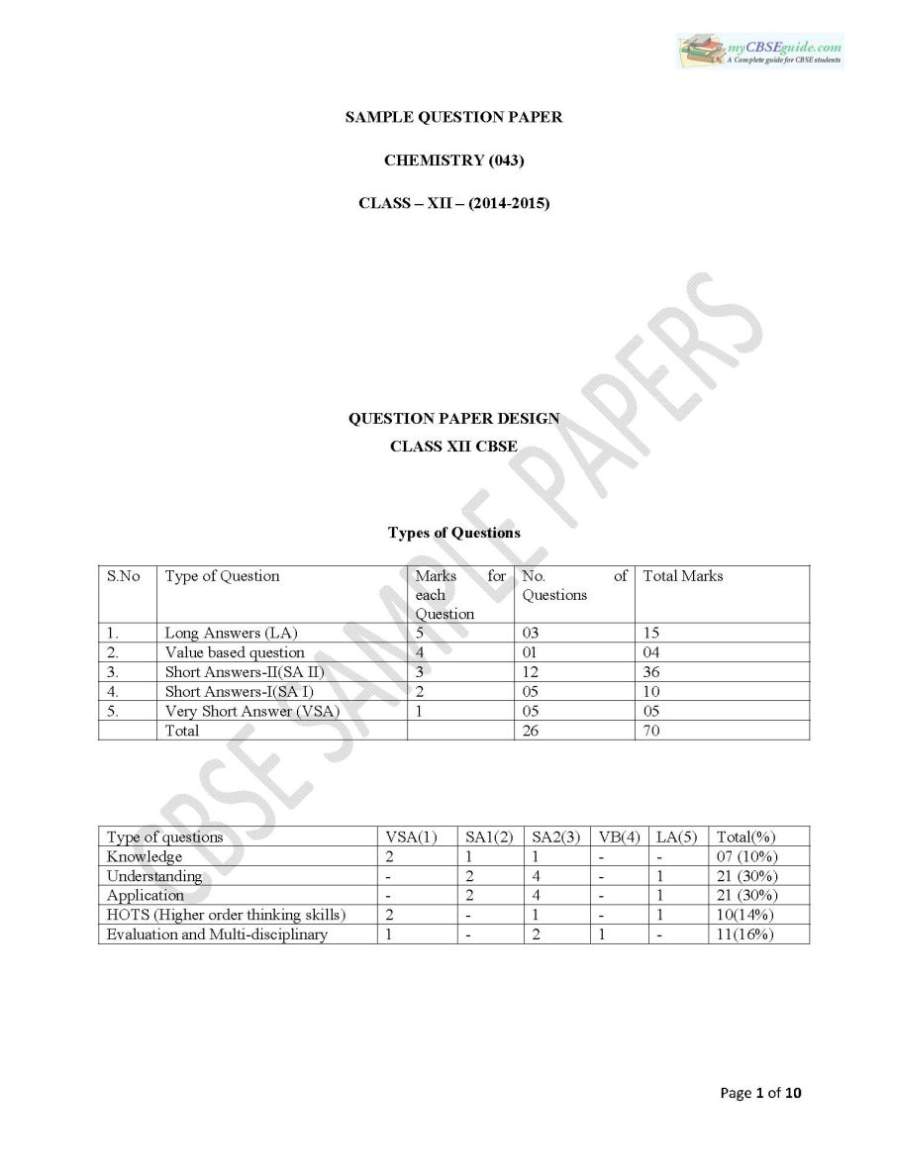     |