|
#3
29th November 2019, 09:22 AM
| |||
| |||
| Re: IITD Chemistry
The details of the Event 2018 - Phosphoric Acid-Catalyzed, Enantioselective Transformations of H-Bonded ortho-Quinone Methides and ortho-Quinone Methide Imines – organised by Department of Chemistry of Indian Institute of Technology Delhi are as follows: Date: August 27th 2018 (Monday) Time: 4 PM Venue: Committee Room, Chemistry Department, 6th Floor Ortho-quinone methides are highly reactive synthetic intermediates which have only recently begun to attract the attention of synthetic organic chemists. They typically react as polarized, electronpoor 1-oxabutadienes with electronrich [2π]-components in hetero-Diels-Alder reactions or with nucleophiles in a conjugate addition both with reconstitution of the aromatic π-system. Using chiral phosphoric acid catalysis (HX) we have been able to generate hydrogen-bonded ortho-quinone methides 1 in situ starting from readily accessible benzhydryl alcohols. A variety of electronrich π-nucleophiles were then added in a conjugate addition event with excellent enantioselectivity. β-Dicarbonyl compounds, enamides, indoles, and naphthols are among the most suitable nucleophiles employed for this purpose and deliver a broad range of benzo-annulated oxygen heterocycles. Furthermore, in a cooperative rhodium-/phosphoric acid-catalyzed approach ortho-quinone methides are trapped with oxonium ylides also generated in situ to yield highly substituted and densely functionalized chromanes. The extension of this strategy to ortho-quinone methide imines 2 and the synthesis of nitrogen heterocycles proceeds likewise with good enantio- and diastereocontrol. References: 1. Reviews: a) W.-J. Bai, J. G. David, Z.-G Feng, M. G. Weaver, K.-L Wu, T. R. R. Pettus, Acc. Chem. Res. 2014, 47, 3655; b) L. Caruana, M. Fochi, L. Bernardi, Molecules 2015, 20, 11733; c) Z. Wang, J. Sun, Synthesis 2015, 47, 3629; d) A. A. Jaworski, K. A. Scheidt, J. Org. Chem. 2016, 81, 10145-10153. 2. a) O. El-Sepelgy, S. Haseloff, S. K. Alamsetti, C. Schneider, Angew. Chem. Int. Ed. 2014, 53, 7923; b) S. Saha, C. Schneider, Chem. – Eur. J. 2015, 21, 2348; c) S. Saha, C. Schneider, Org. Lett. 2015, 17, 648; d) S. Saha, S. K. Alamsetti, C. Schneider, Chem. Commun 2015, 51, 1461 3. S. K. Alamsetti, M. Spanka, C. Schneider, Angew. Chem. Int. Ed. 2016, 55, 2392 4. a) M. Kretzschmar, T. Hodik, C. Schneider, Angew. Chem. Int. Ed. 2016, 55, 9788, b) T Hodik, C. Schneider, Org. Biomol Chem. 2017, 15, 3706-3716. 5. M. Kretzschmar, F. Hofmann, D. Moock, C. Schneider, Angew. Chem. Int. Ed. 2018, 57 4774 Event 2018 - Phosphoric Acid-Catalyzed, Enantioselective Transformations of H-Bonded ortho-Quinone Methides and ortho-Quinone Methide Imines –Department of Chemistry of IIT Delhi  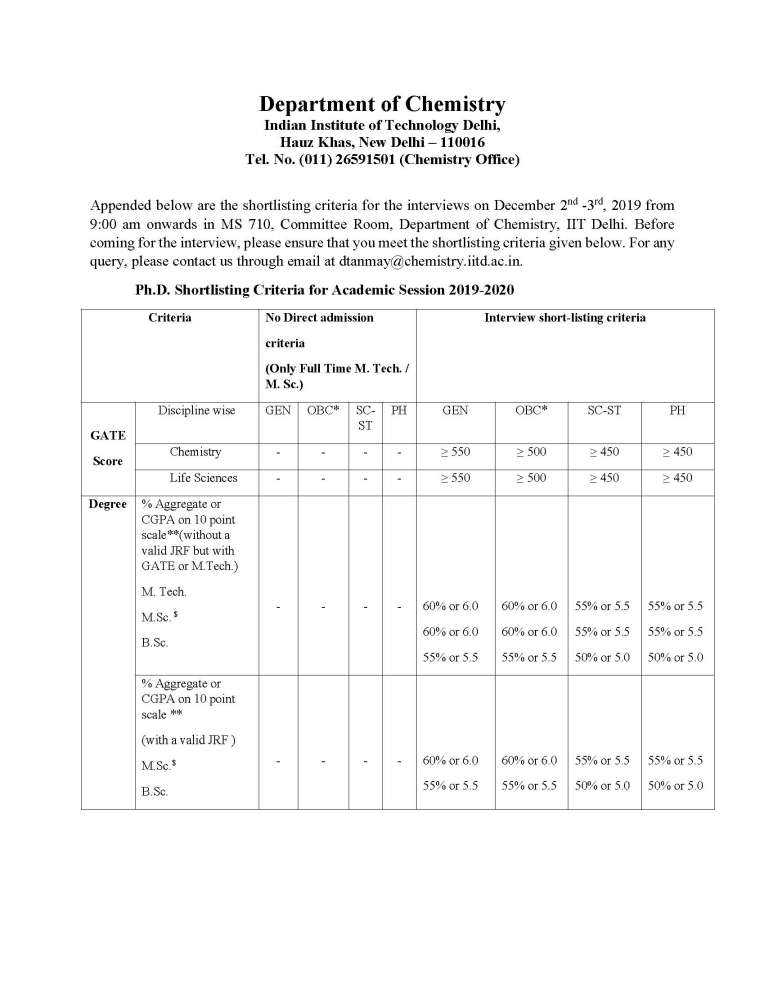   |