|
#2
20th February 2015, 03:55 PM
| |||
| |||
| Re: model question paper pcmb NCERT
Hello friend as you want the Model Question Papers of Physics, Chemistry, Mathematics, Biology so here I am providing you the same…. 1. The question paper has four parts: A, B, C and D. All parts are compulsory. 2. Write balanced chemical equations and draw labelled diagrams wherever required. 3. Use log tables and the simple calculators if necessary. (Use of Scientific Calculators is not allowed) PART- A I. Answer ALL of the following. ( Each questions carries 1 mark) 10 × 1 = 10 1. State Henry’s law. 2. What is the conclusion drawn when Vant Hoff’s factor of a solution is less than one? 3. How molar conductivity varies with dilution? 4. Mention one criterion for intermolecular collisions of two reactants to be effective. 5. Name the metal that is refined by Van Arkel method. 6. Name the first noble gas compound prepared by Neil Bartlett? 7. Give an example for Heteroleptic complex. 8. Write the IUPAC name for CH3-CH(CH3)-CH2Cl. PART- B II. Answer Any FIVE of the following. ( Each questions carries 2 mark) 5 × 2 = 10 11. a) Give one example for paramagnetic substance. b) Which type of binding force existing in ice? 12. Write anodic and cathodic half-cell reactions taking place in Daniel cell. 13. Show that for first order reaction t87.5% = 3 t50%. 14. What is lanthanide contraction? What is the cause for it? 15. How do you convert bromoethane into tertiary-butylethyl ether? Give the chemical equation of the reaction. 16. What is Stephen’s reaction? Give the chemical equation of the reaction. 17. What is the therapeutic action of a) Paracetamol b) Barbituricacid. 18. What are anionic detergents? Give an example. PART- C III. Answer Any FIVE of the following. ( Each questions carries3 mark) 5 × 3 = 15 19. Explain the extraction of aluminium from purified alumina by Hall-Heroult process. 20. Explain manufacture of nitric acid by Ostwald’s process. 21. a) How is ozonised oxygen prepared? b) Write the structure of sulphurous acid. 1 22. a) How does phosphorus react with limited amount of chlorine? Give equation. b) What is the product obtained by the reaction between fluorine in excess of bromine? 23. a) 3d Transition metals and their compounds are good catalysts. Give two reasons? b) Give the formula for the calculation of spin only magnetic moment. 24. Explain the manufacture of Potassium dichromate from chromite ore. 25. Using VBT explain, i) Geometry of the complex ii) Type of hybridization, in the complex ion [CoF6] 3-. iii) Is it an inner or outer orbital complex? 26. a) Draw the structures of cis -trans isomers for [Pt (NH3)2.Cl2]. b) How many ions are produced from the aqueous solution of complex K3[Al(C2O4)3] PART –D IV. Answer Any THREE of the following. ( Each questions carries5 mark) 3 × 5 = 15 27. a) Calculate the packing efficiency in a Face Centered Cubic lattice. 3 b) If a metal with atomic mass 209 crystallizes in a simple cubic lattice what is the edge length of its unit cell. (Given d = 91.5 kg m-3). 2 28. a) 5.8g of a non-volatile solute was dissolved in 100g of carbon disulphide (Molar mass = 76). The vapour pressure of the solution was found to be 190mmHg. Calculate the molecular mass of the solute. The vapour pressure of pure carbon disulphide 195 mm Hg. 3 b) What are azeotropes? Give an example. 29. a) The resistance of M/10 solution is found to be 2.5 X 103 ohms. Calculate molar conductance (Given Cell constant = 1.15 cm-1). 3 b) Mention two general methods for prevention of corrosion. 2 30. a) Derive an expression for rate constant of a first order reaction. b) What is the effect of positive catalyst on energy of activation? 1 31. a) Mention two factors which affect adsorption of a gas on a solid. b) Is synthesis of ammonia by Haber’s process, a homogeneous or heterogeneous catalysis? c) State Hardy-Schulze rule. Among Al2(SO4)3 and (NH4)3PO4 which is better coagulating agent for a negative sol ? 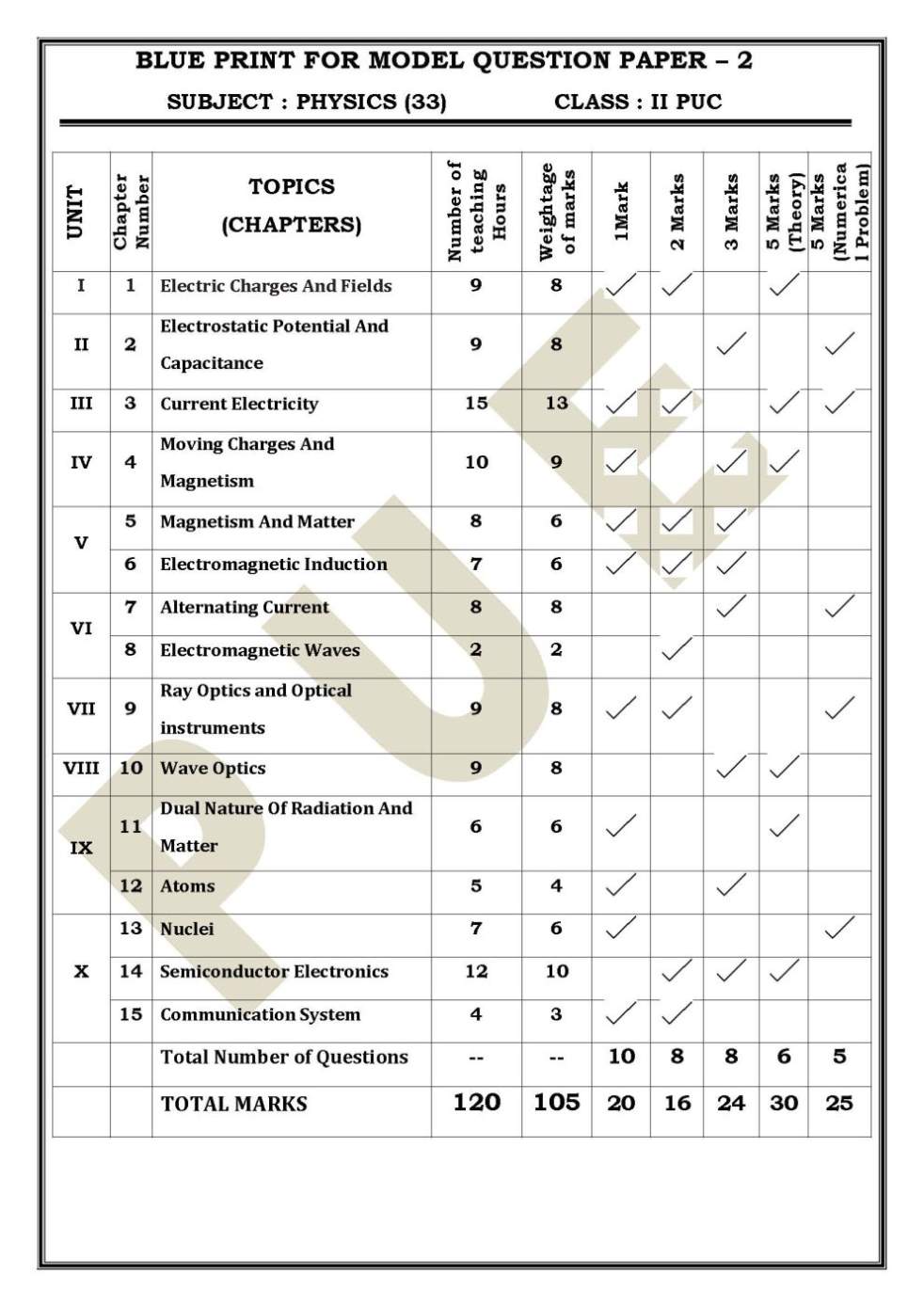   For more papers here I am attaching a PDF files please download it… Last edited by sumit; 13th December 2019 at 02:57 PM. |




