|
#4
23rd May 2015, 02:39 PM
| |||
| |||
| Re: IIT Study Material Chemistry
Here you want to get last years question papers of IIT JEE Chemistry, so here I am giving following question papers to you: IIT JEE Chemistry Question Paper I Among cellulose, poly(vinyl chloride), nylon and natural rubber, the polymer in which the intermolecular force of attraction is weakest is (A) Nylon (B) Poly(vinyl chloride) (C) Cellulose (D) Natural R The compound(s) formed upon combustion of sodium metal in excess air is(are) (A)Na2O2 (B)a2O (C)NaO2 (D)NaOH The correct statement(s) about the compound H3C(HO)HC−CH=CH− CH(OH)CH3(X) is(are) (A) The total number of stereoisomers possible for X is 6 (B) The total number of diastereomers possible for X is 3 (C) If the stereochemistry about the double bond in X is trans, the number of enantiomers possible for Xis 4 (D) If the stereochemistry about the double bond in X is cis , the number of enantiomers possible for X is 2 The correct statement(s) rega rding defects in solids is(are) (A) Frenkel defect is usually favoured by a very small difference in the sizes of cation and anion (B) Frenkel defect is a dislocation defect (C) Trapping of an electron in the lattice leads to the formation of F-center (D) Schottky defects have no effect on the physical properties of solids IIT JEE Chemistry Question Paper I  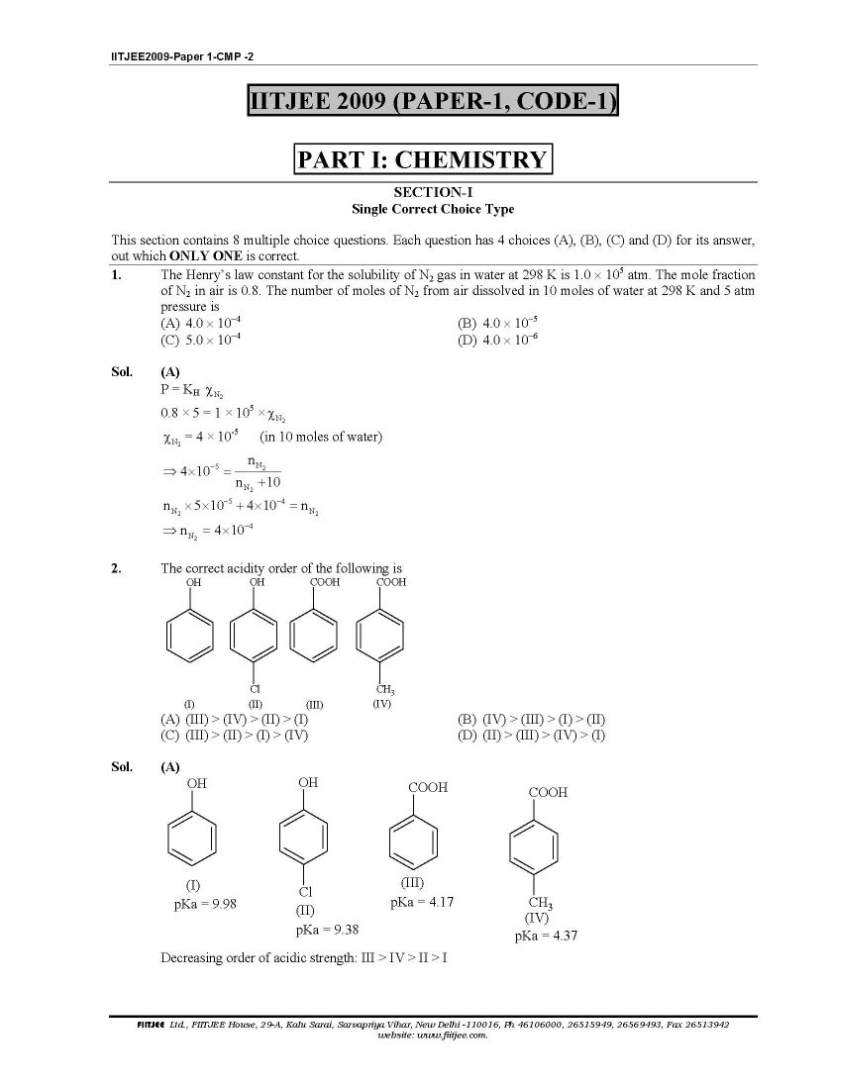   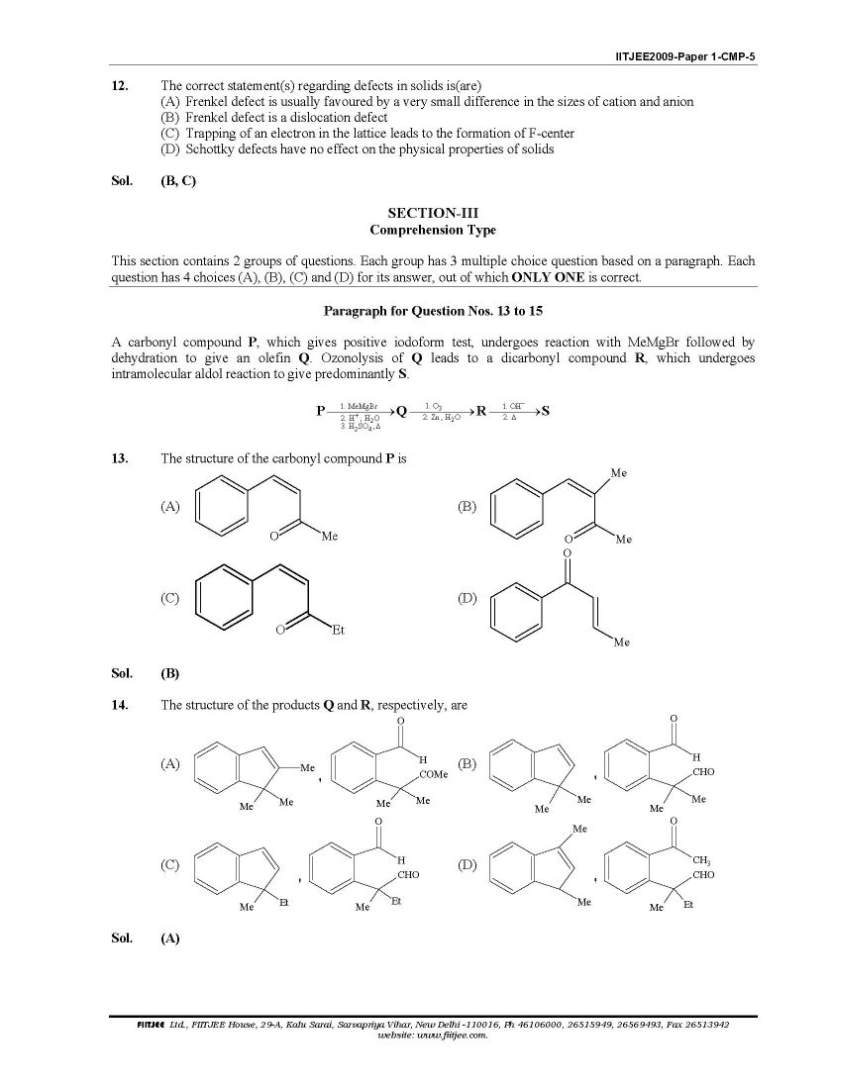 more papers detail to attached two pdf files; |