|
#2
18th November 2015, 03:13 PM
| |||
| |||
| Re: Iit qp
As you want I am here giving you sample Question Paper for IIT JEE examination. Sample paper : 21. Which ordering of compounds is according to the decreasing order of the oxidation state of nitrogen? (A) HNO3, NO, NH4Cl, N2 (B) HNO3, NO, N2, NH4Cl (C) HNO3, NH4Cl, NO, N2 (D) NO, HNO3, NH4Cl, N2 As per IUPAC nomenclature, the name of the complex [Co(H2O)4(NH3)2]Cl3 is : (A) Tetraaquadiaminecobalt (III) chloride (B) Tetraaquadiamminecobalt (III) chloride (C) Diaminetetraaquacoblat (III) chloride (D) Diamminetetraaquacobalt (III) chloride Ans. (D) Sol. [Co(H2O)4 (NH3)2 ]Cl3 = Diamminetetraaquacobalt (III) chloride. 29. The carboxyl functional group ( ˇ (A) picric acid (B) barbituric acid (C) ascorbic acid (D) aspirin The colour of light absorbed by an aqueous solution of CuSO4 is : (A) organge-red (B) blue-green (C) yellow (D) violet Choose the correct reason(s) for the stability of the lyophobic colloidal particles. (A) Preferential adsorption of ions on their surface from the solution. (B) Preferential adsorption of solvent on their surface from the solution. (C) Attraction between different particles having opposite charges on their surface. (D) Potential difference between the fixed layer and the diffused layer of opposite charges around the colloidal particles. Ans. (AD) Sol. (A) due to preferential adsorption of common ions (B)X (C) X (due to repulsion not due to attraction) (D) The layer of oppositely charged particles around any colloidal particles will decrease the potential energy of system as a whole. IIT JEE exam paper 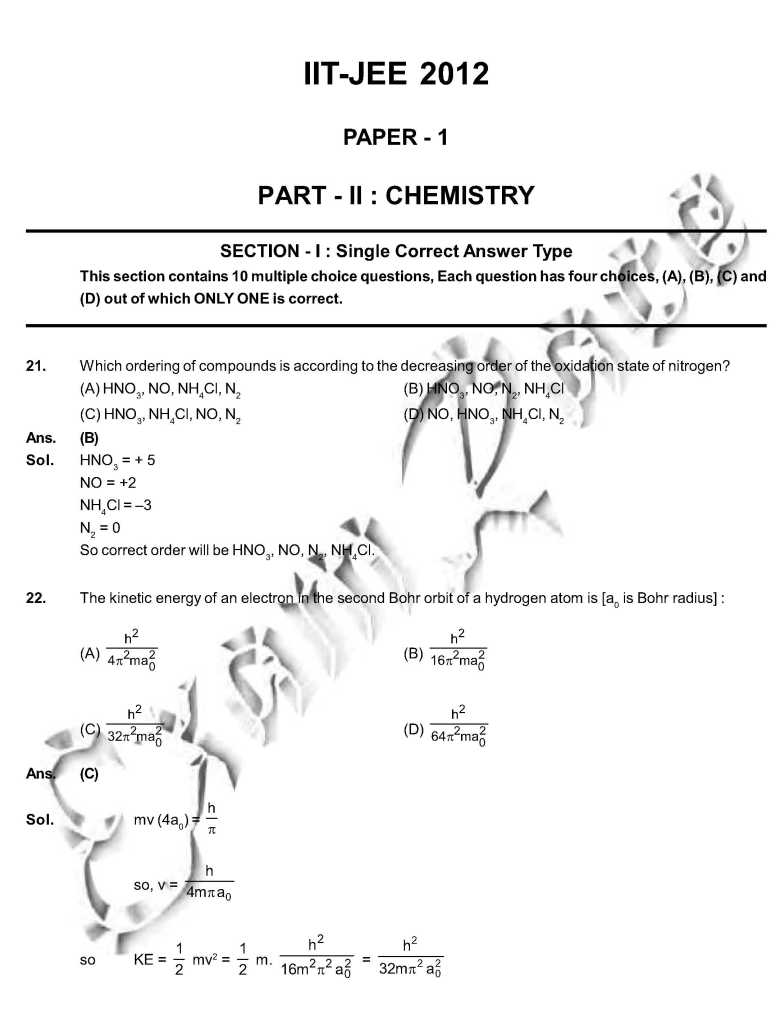 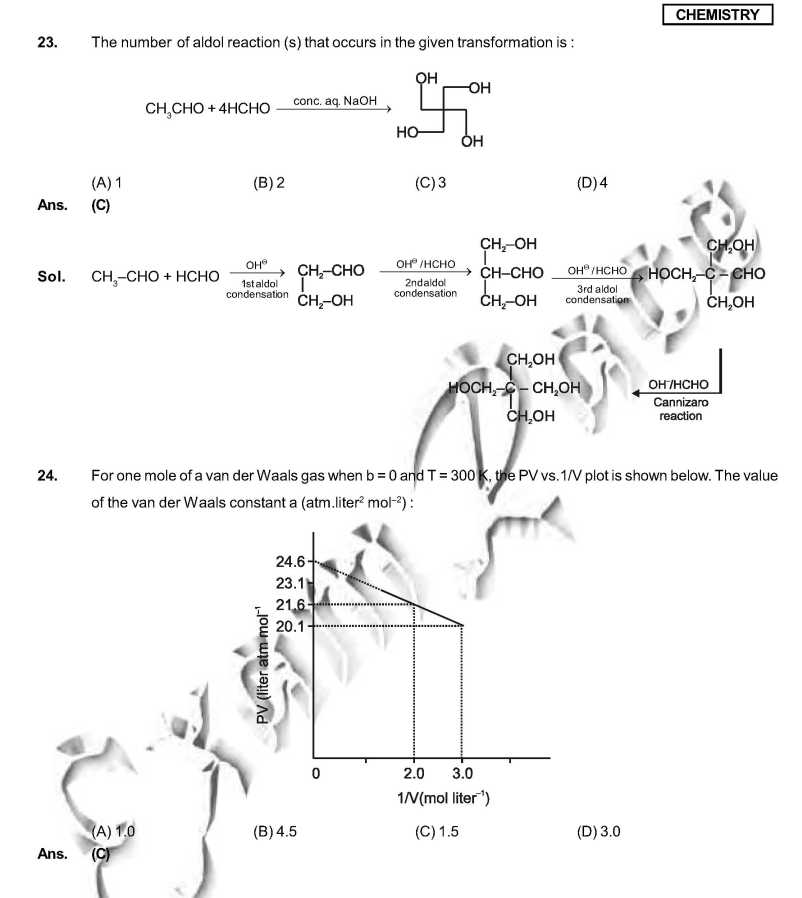  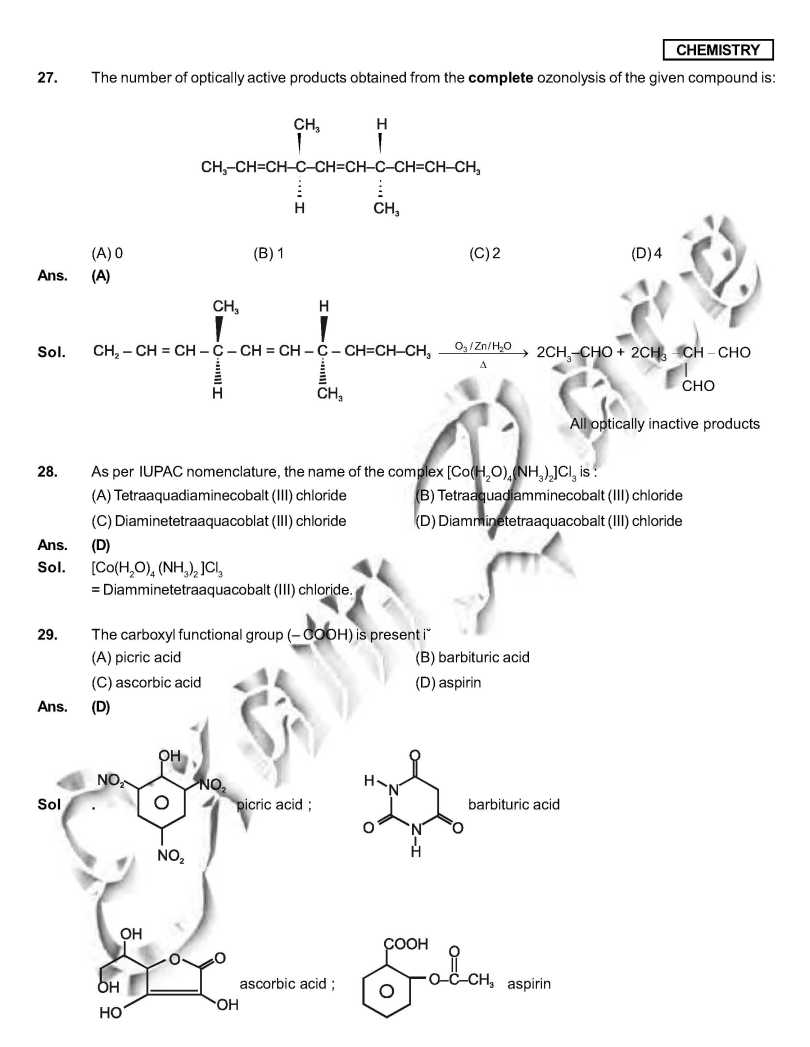 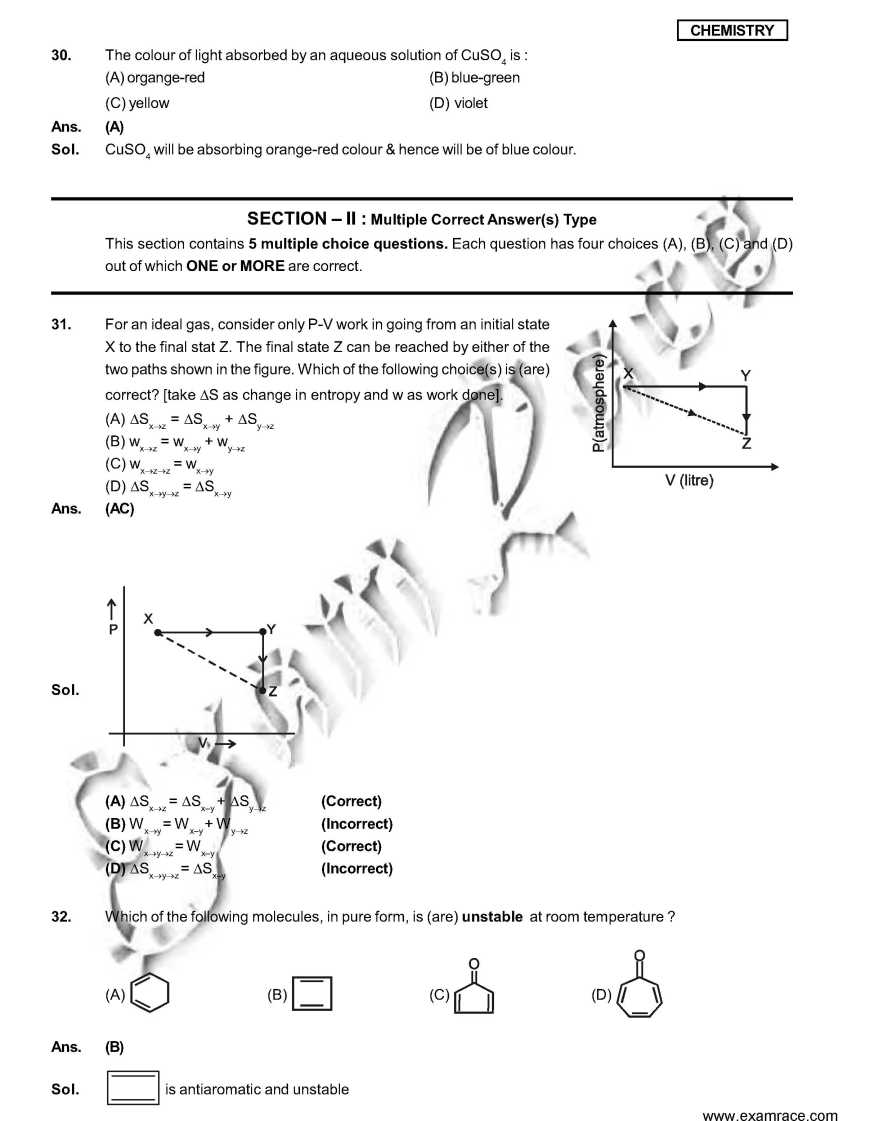 Here is the attachment. |