|
#3
10th July 2018, 09:39 AM
| |||
| |||
| Re: Bharathiar University Chemistry Syllabus
As you want here I am giving below Bharathiar University M.Sc. Chemistry program syllabus on your demand : ORGANIC CHEMISTRY I ( Reaction Mechanisms ) Course Code: CHMA13A Number of credit hours : 4 (four) Subject Description: This course presents the principles of various organic reactions and outlines the mechanism and discusses the applications of reactions Goals: To enable the students to learn the principles of reaction mechanism and modern reagents used for various reactions Objectives On successful completion of the course the students should have i) Understood the principles and reaction mechanism involving various electrophonic, nucleophilic, addition, elimination, redox reactions & molecular rearrangements: Contents Unit I Aliphatic and Aromatic Nnucleophilic Substitution Reactions: Bonding - structure and reactivity - acids and bases (hard and soft acid base theory) - methods of determination and the study of reaction mechanisms. SN 1 , SN 2 , SN i and neighbouring group mechanisms - kinetics - effects of structure - solvent and leaving and entering group - stereochemistry - hydrolysis of esters - Wurtz reaction - Claisen and Dieckmann condensation - Williamson reactions. Different mechanisms of aromatic nucleophilic substitution - Ziegler alkylation - Chichibabin reaction - cine substitution - diazonium group as leaving group Unit - II Aliphatic and Aromatic Electrophilic Substitution Reactions: SE 1 and SE 2 reactions - mechanisms and reactivity - typical reactions involving migration of double bond - keto-enol tautomerism - halogenation of carbonyl compounds - Stork enamine reactions - decarboxylation of aliphatic acids - Friedel Crafts acylation of olefinic carbon. Aromatic electrophilic substitution - reactivity - orientation and mechanisms - nitration - halogenation and sulphonation - Friedel Crafts alkylation - Friedel Crafts arylation (Scholl reaction) and acylation - Jacobsen reaction - formylation with (i) disubstituted formamides(Vilsmeyer- Haack reaction) (ii) zinc cyanide and HCl (Gattermann reaction) (iii) chloroform (Reimer - Tiemann reaction) - carboxylation with (i) carbonyl halides (ii) carbon dioxide (Kolbe Schmidt reaction) - amidation with isocyanates - hydroxyalkylation (hydroxyalkyl - dehydrogenation)- cyanodehydration of aldehydes and ketones (Bradsher reaction and Bischler - Napieralski rection) - haloalkylation - aminoalkylation and amido alkylation - thioalkylation -acylation with nitriles (Hoesch reaction) - cyanation - hydroxylation. Unit III Molecular Rearrangements: Molecular rearrangements - intramolecular rearrangements - 1,2- shifts in carbonium ions - Wagner-Meerwein and related rearrangements - Demjanov rearrangement - migration to carbonyl carbon - Neber rearrangement Benzilic acid- Baeyer-Villiger rearrangement - rearrangements to electron deficient nitrogen and oxygen - dienone-phenone - Favorski - Wolf - benzidine - Claisen - Cope rearrangement, Ylides: Stevens-Wittig-Sommelet-Gruvenstein- Zimmermann rearrangements- non-cyclic rearrangements - Chapman - Wallach rearrangement Bharathiar University M.Sc. Chemistry program syllabus 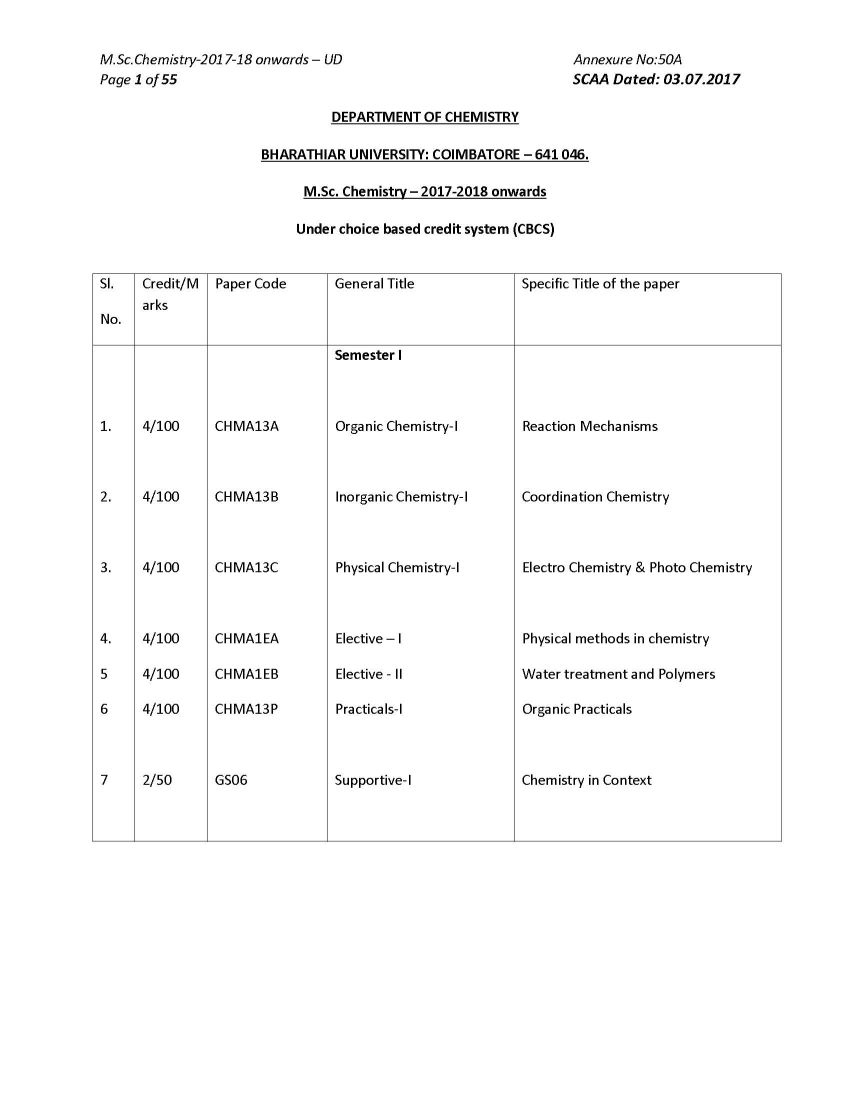 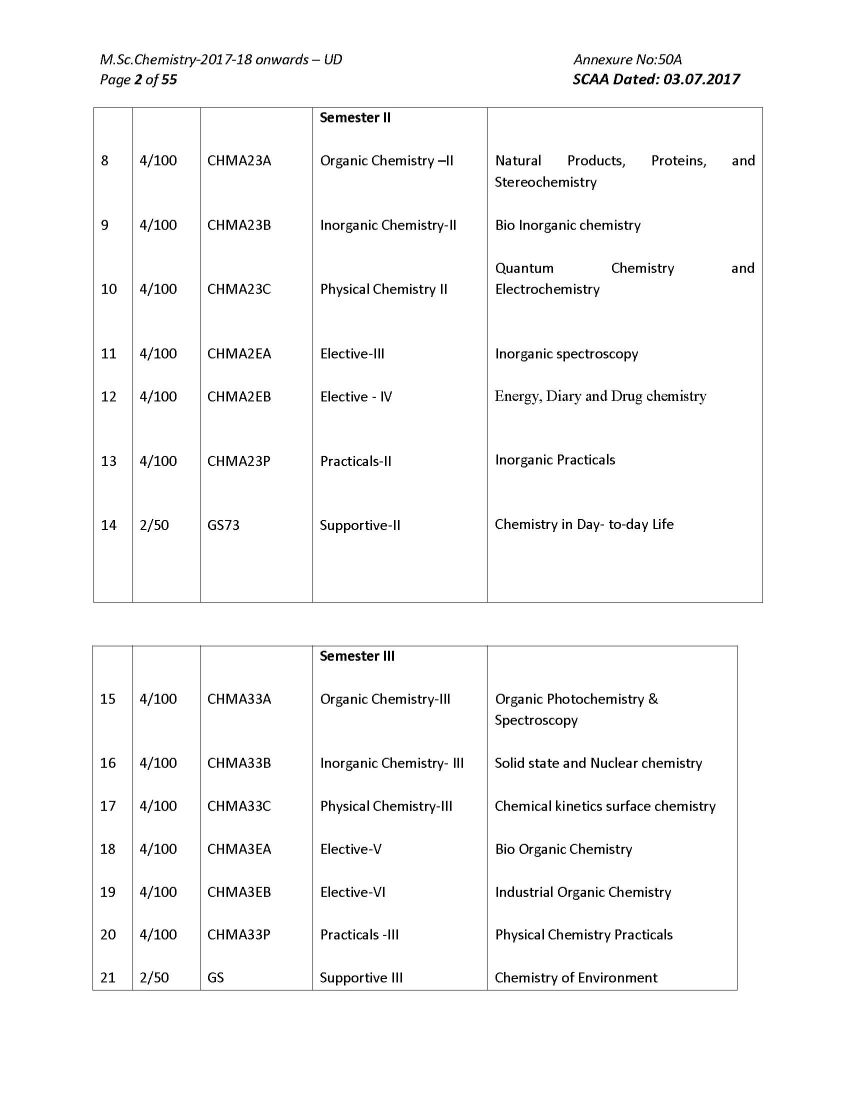 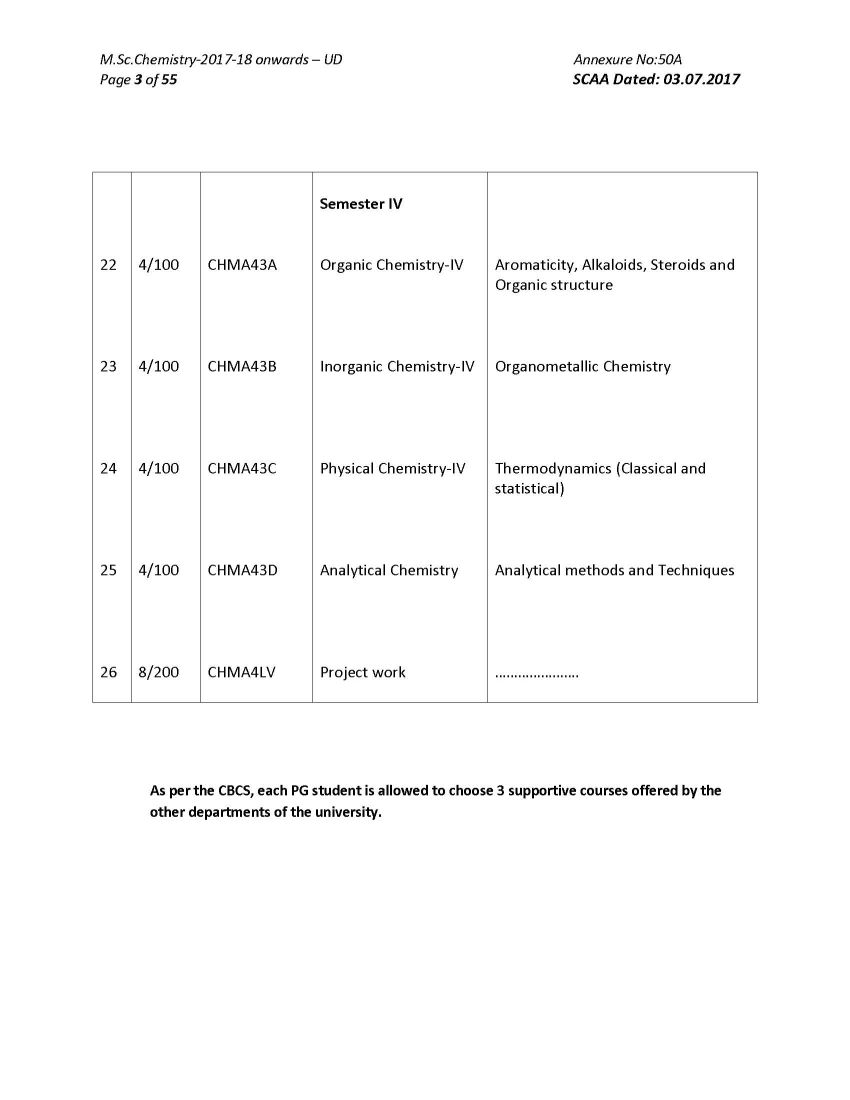  |