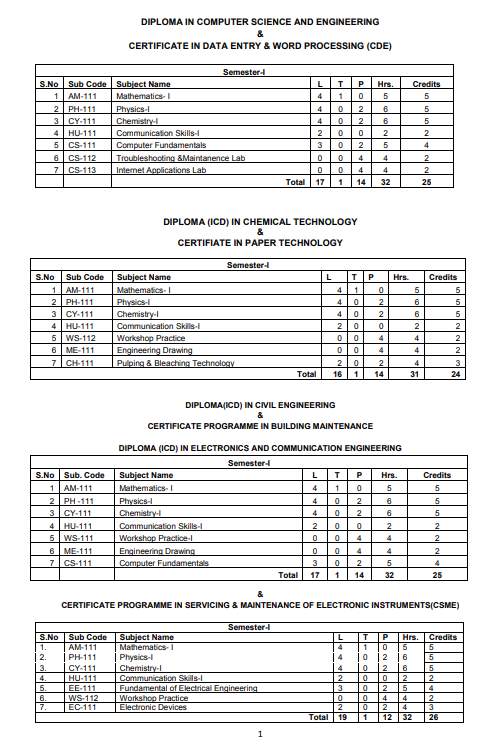
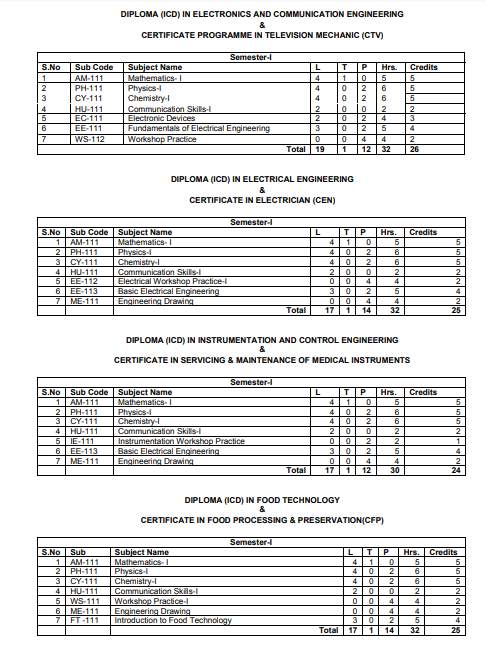
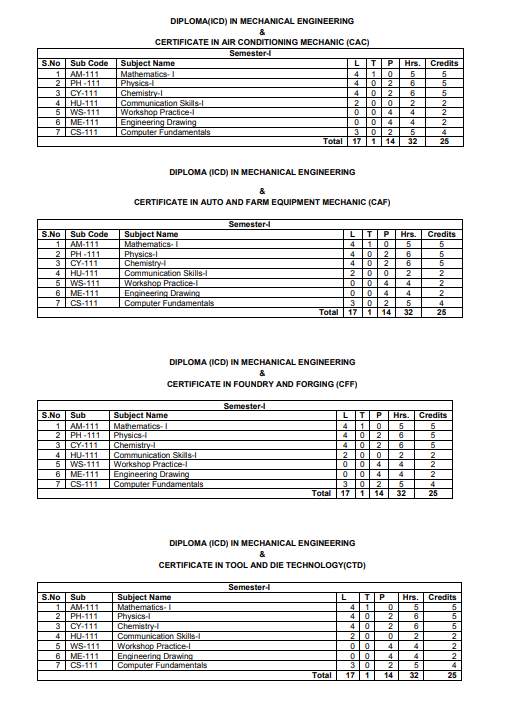
The ICD 1st Sem Syllabus offered by SLIET (Sant Longowal Institute of Engineering & Technology, Longowal) is as follows:
Title of the course : CHEMISTRY-I
Subject Code : CY-111
Credit : 5 (Lecture 4; Practical 1)
Course Outlines
Unit I Atomic Structure
Introduction to atom and its constituent particles, Bohr’s model of atom, Line spectrum of hydrogen, Dual nature of radiation, de Broglie’s relationship, Uncertainty principle, Quantum numbers, Shapes of orbitals, Pauli’s exclusion principle, Aufbau Energy ranking rule, Hund’s rule, Electronic configuration of atoms
Modern periodic table
Modern periodic table, Periodic properties (ionization potential, electron affinity, atomic and ionic radii), Variation of periodic properties along a period and group, Introduction to s and pblock elements
Chemical Bonding
Types of chemical bond (ionic, covalent, cooordinate), Lewis structure, Valence bond theory,
VSEPR theory, Hybridization, Molecular orbital theory of homonuclear diatomic molecules,
Vanderwaal forces, Hydrogen bond, Metallic bond
Chemical and Ionic Equilibrium
Law of chemical equilibrium, Le Chatelier’s principle, Law of mass action, Equilibrium constant, Ionic equilibrium – ionization of acids and bases, Strong and weak electrolytes, Degree of ionization, Concept of pH, Hydrolysis of salts, Common Ion effect and Solubility product, Concept of acids and bases, Buffer solutions
Unit II Chemical Thermodynamics
Concepts of extensive and intensive properties, State functions, First law of Thermodynamics:
Internal energy, Enthalpy, Heat capacity and Specific heat, Applications of First law of thermodynamics, Hess’s law of constant heat summation, Second law of thermodynamics: Entropy, Free energy, Spontaneity of a chemical reaction, Free-energy change and Chemical equilibrium
Organic Chemistry
Classification and IUPAC nomenclature of organic compounds, Inductive effect, Electromeric effect, Resonance and Hyperconjugation, Electrophiles and Nucleophiles Reaction
Intermediates - carbocations, carbanions, free radicals, Types of organic reactions, Stereoisomerism: Optical, Geometrical and Conformational
Environmental chemistry
Environmental pollutants: soil, water and air pollution, Chemical reactions in atmosphere,
Kinds of smog, Major atmospheric pollutants, Acid rain, Ozone and its reactions, Effects of the depletion of ozone layer, Greenhouse effect and Global warming – Industrial air pollution, Green chemistry as an alternative tool for reducing pollution
ICD 1st Sem Syllabus Sant Longowal Institute of Engineering & Technology, Longowal